Functional and Ultrasound Guided Resection of Glioblastoma - FUTURE-GB
Glioblastoma (GB) is the most common primary brain tumour and is incurable. It grows very quickly from the brain tissue itself, rather than from a cancer elsewhere in the body. It is expected that the number of people with a brain tumour will rise by 6% in the UK between 2014 and 2035. However, prognosis (outcome) remains extremely poor, with most people surviving just over 12 months, and as a patient’s tumour grows patients experience a reduction (decline) in their quality of life. Therefore, we need to ensure quality of life, which remains difficult. The main treatments for GB are surgery, radiotherapy and chemotherapy, given in combination.
For patients where it is thought that surgery will benefit, a surgeon often removes as much tumour as possible, whilst limiting the risk of causing damage, such as weakness, speech, or cognitive difficulties. However, which technology a surgeon should use during surgery to remove the tumour safely is unclear. This can affect how soon the cancer returns, what effects of surgery or symptoms a patient develops, and how a patient feels.
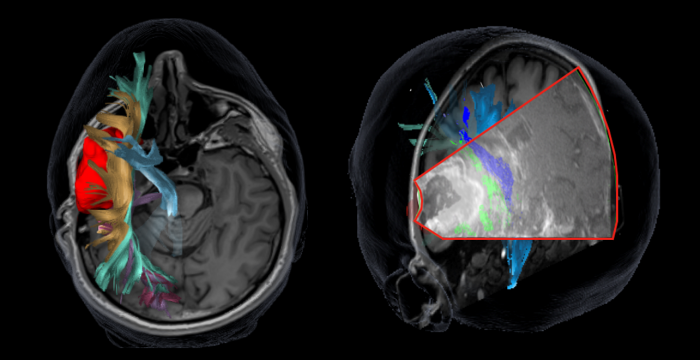
High frequency sound waves that create an image, called Ultrasound (US), is one of the tools a surgeon can use during the operation to find the tumour and see how much is removed. Another technology, Diffusion Tensor Imaging (DTI), allows important nerve pathways involved in certain functions, for example, speech/language, vision and movement, to be avoided in surgery.
This trial aims to see if GB surgery with these extra technologies (tools) added to the standard ones, increases a patient’s good functioning quality of life, so-called Deterioration Free Survival (DFS).